40 lewis dot diagram of ammonia
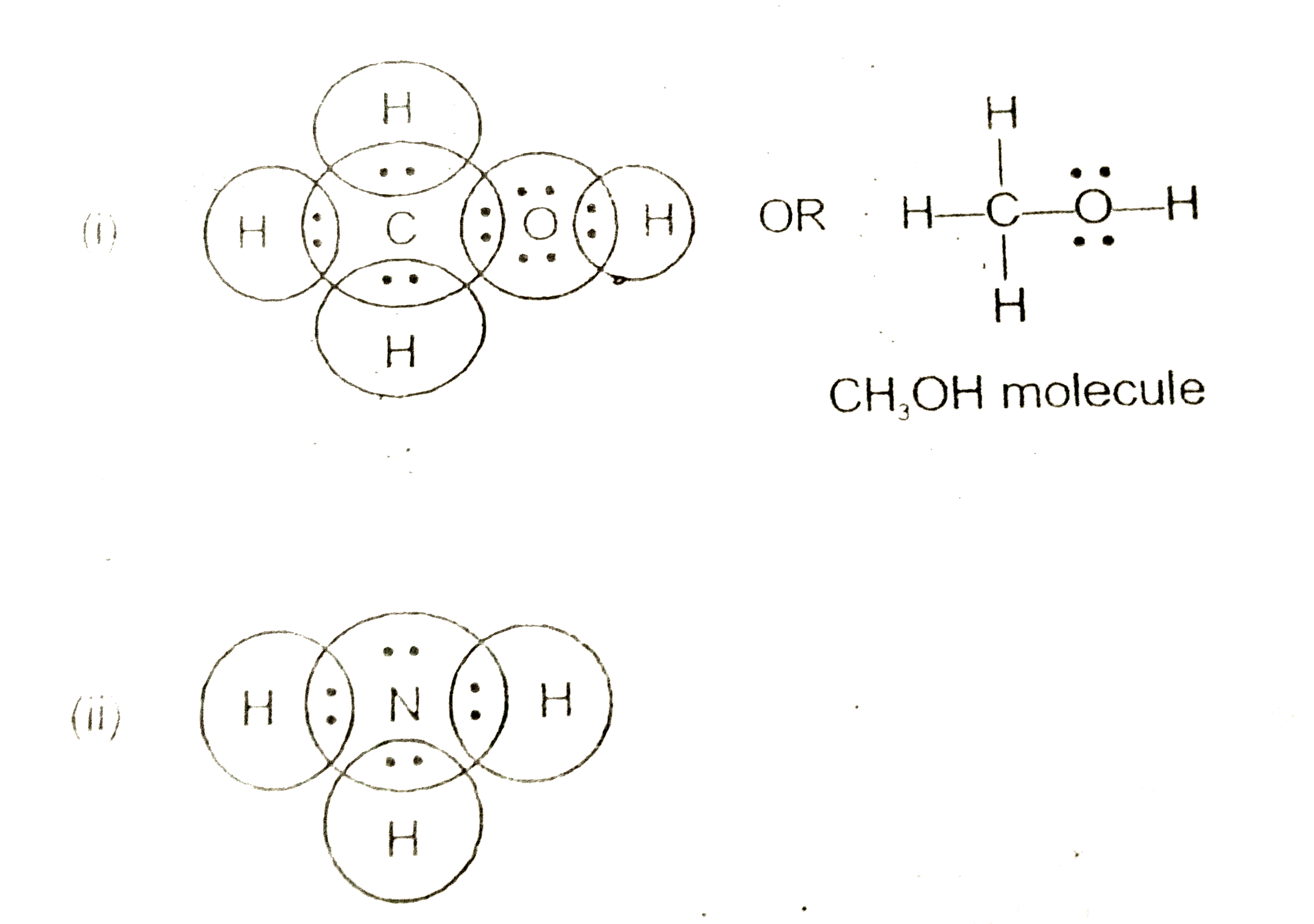
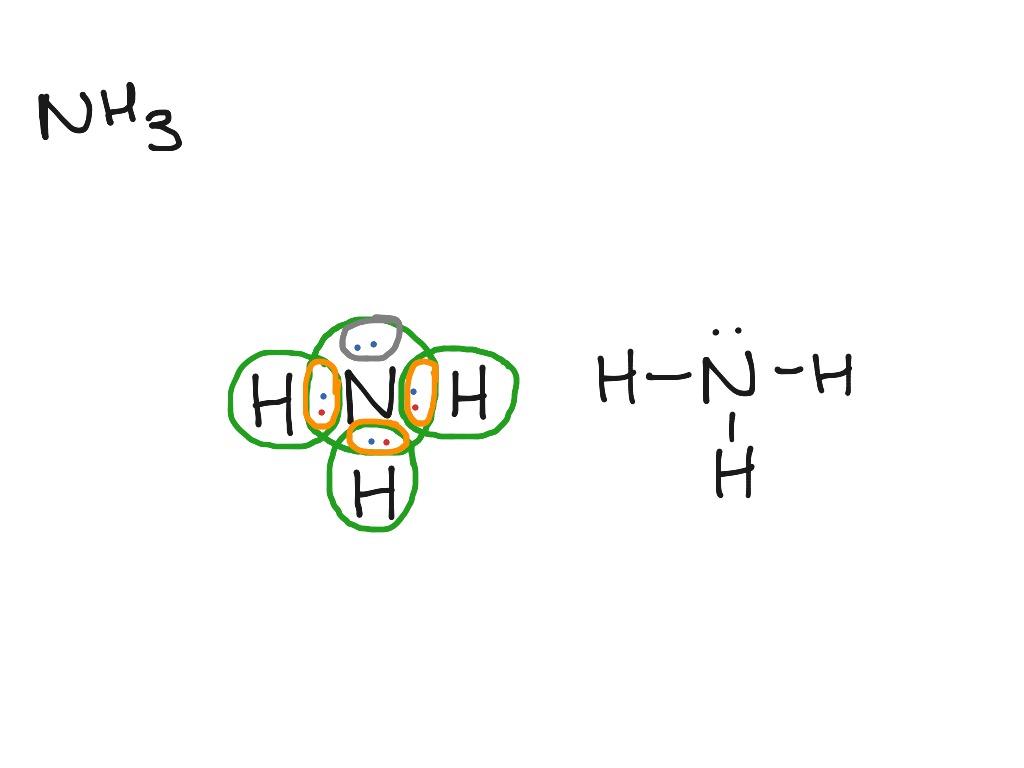
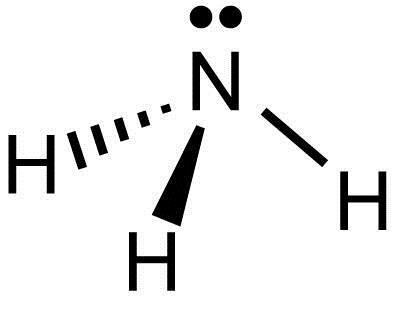
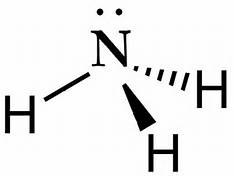
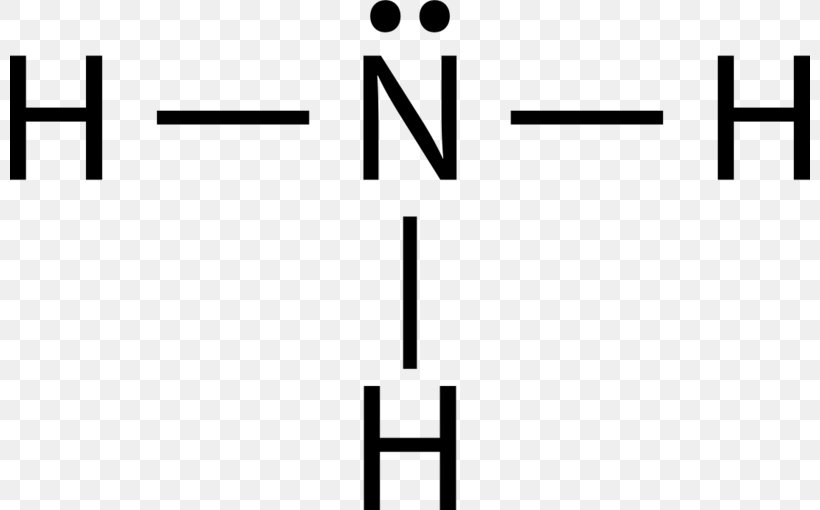
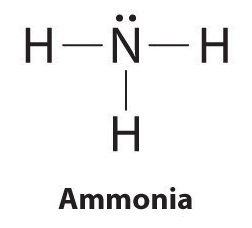
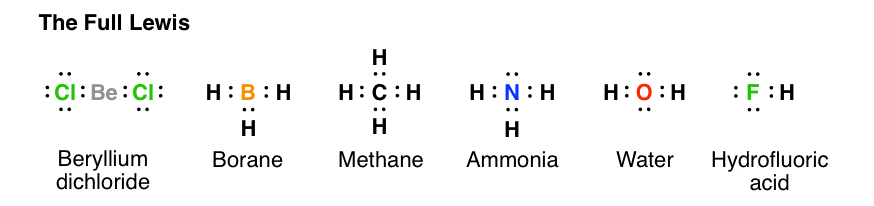
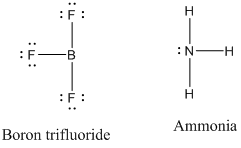
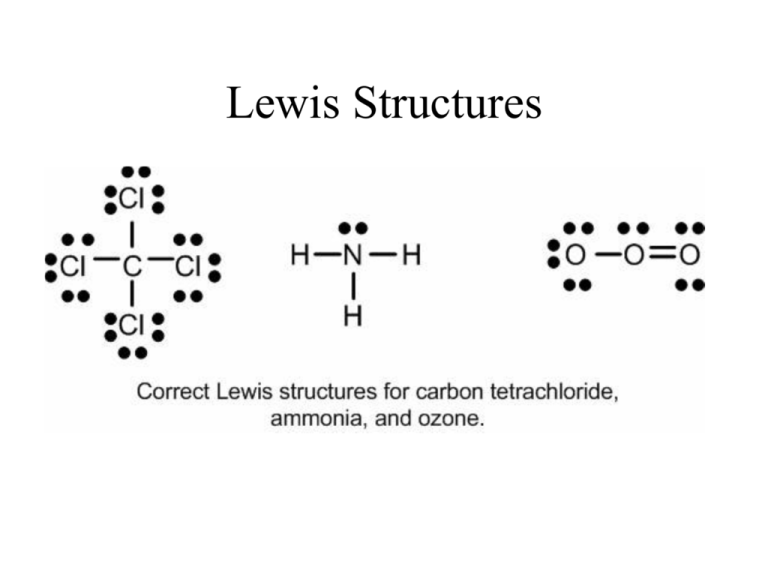
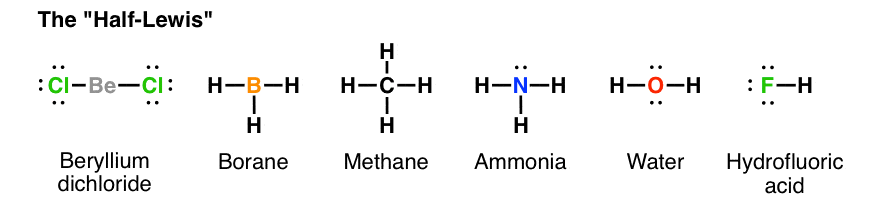
Drawing the Lewis Structure for NH 3 ( Ammmonia) Ammonia (NH 3) is a commonly tested Lewis structure due to it's widespread use in agriculture as a fertilizer. It also is a good example of a molecule with a trigonal prymidal molecular geometry. There are 8 valence electrons available for the Lewis structure for NH 3. What is the Lewis dot structure for ammonia? In the lewis structure of ammonia (NH3), there are three N-H bonds and one lone pair on nitrogen atom. Lewis structure of NH3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Can you draw the electron dot structure of ammonia?
What is the Lewis dot diagram of NH3? The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons. What is the structure for NH3? NH3.

Lewis dot diagram of ammonia
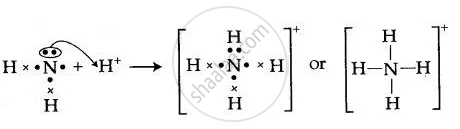
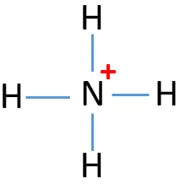
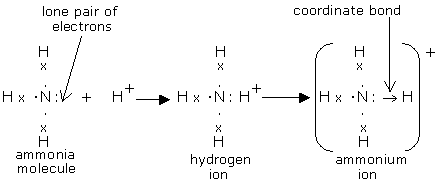
In the lewis structure of ammonia (NH 3), there are three N-H bonds and one lone pair on nitrogen atom. Lewis structure of NH 3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Each step of drawing the lewis structure of NH 3 is explained in detail in this tutorial. Can I draw the NH 4 + lewis structure from NH 3 lewis structure? First, we need the reaction of forming of NH4+ ion from NH 3. NH 4 + ion is made from NH 3 and H + ion. The lone pair in NH 3 is given to the H + ion to make a new N-H bond. Now, there are four N-H bonds around nitrogen atom. See the reaction of ammonia and HCl. Nov 29, 2018 · Lewis Dot Diagram Of Nh3. The Lewis structure of ammonia, NH3, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons. Electron Dot Structure of NH3 by Jeff Bradbury - February 17, - Lewis Electron Dot Structure for ammonia molecule NH3. by crator-avatar Jeff Bradbury 2.
Lewis dot diagram of ammonia. A step-by-step explanation of how to draw the NH3 Lewis Dot Structure (Ammonia).For the NH3 structure use the periodic table to find the total number of vale... The molecular weight of Ammonia is 17 g/mol. It is a colourless alkaline gas. Starting with the Lewis dot structure of Ammonia, Nitrogen has 5 valence electrons and each hydrogen has 1 valence electron. So, the total valence electrons are 8. Hydrogen always goes on the outside, so Nitrogen is the central atom. A simple method for drawing the Lewis structure for ammonia. Dec 27, 2018 · Use information from step 4 and 5 to draw the lewis structure. Nitrogen goes in the centre. Lewis dot structure of ammonia. Alternatively a dot method can be used to draw the lewis structure of NH3. Calculate the total valence electrons in NH3 molecule. N=5,H=1x3=3 Total=8 Put Nitrogen in the center and three hydrogen atoms on the sides. Lewis Dot Structures (2): Water and Ammonia. Drawing the Lewis Structure for NH 3. Viewing Notes: NH 3 (Ammonia) is a commonly tested Lewis structure.
Nov 29, 2018 · Lewis Dot Diagram Of Nh3. The Lewis structure of ammonia, NH3, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons. Electron Dot Structure of NH3 by Jeff Bradbury - February 17, - Lewis Electron Dot Structure for ammonia molecule NH3. by crator-avatar Jeff Bradbury 2. Can I draw the NH 4 + lewis structure from NH 3 lewis structure? First, we need the reaction of forming of NH4+ ion from NH 3. NH 4 + ion is made from NH 3 and H + ion. The lone pair in NH 3 is given to the H + ion to make a new N-H bond. Now, there are four N-H bonds around nitrogen atom. See the reaction of ammonia and HCl. In the lewis structure of ammonia (NH 3), there are three N-H bonds and one lone pair on nitrogen atom. Lewis structure of NH 3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Each step of drawing the lewis structure of NH 3 is explained in detail in this tutorial.









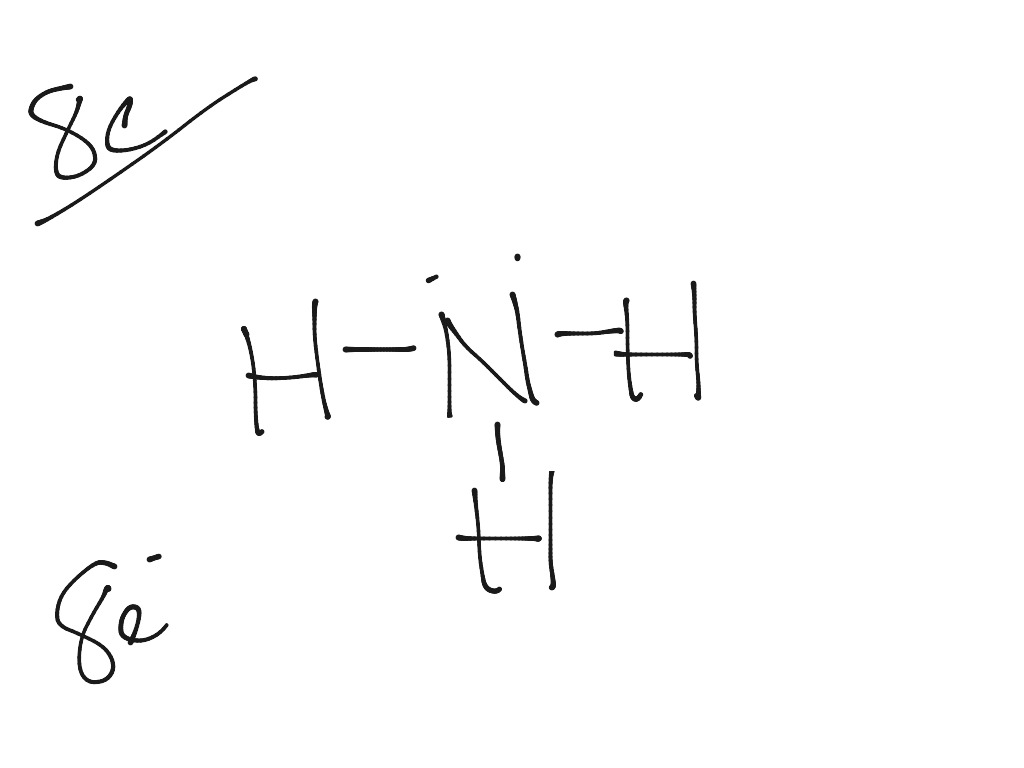

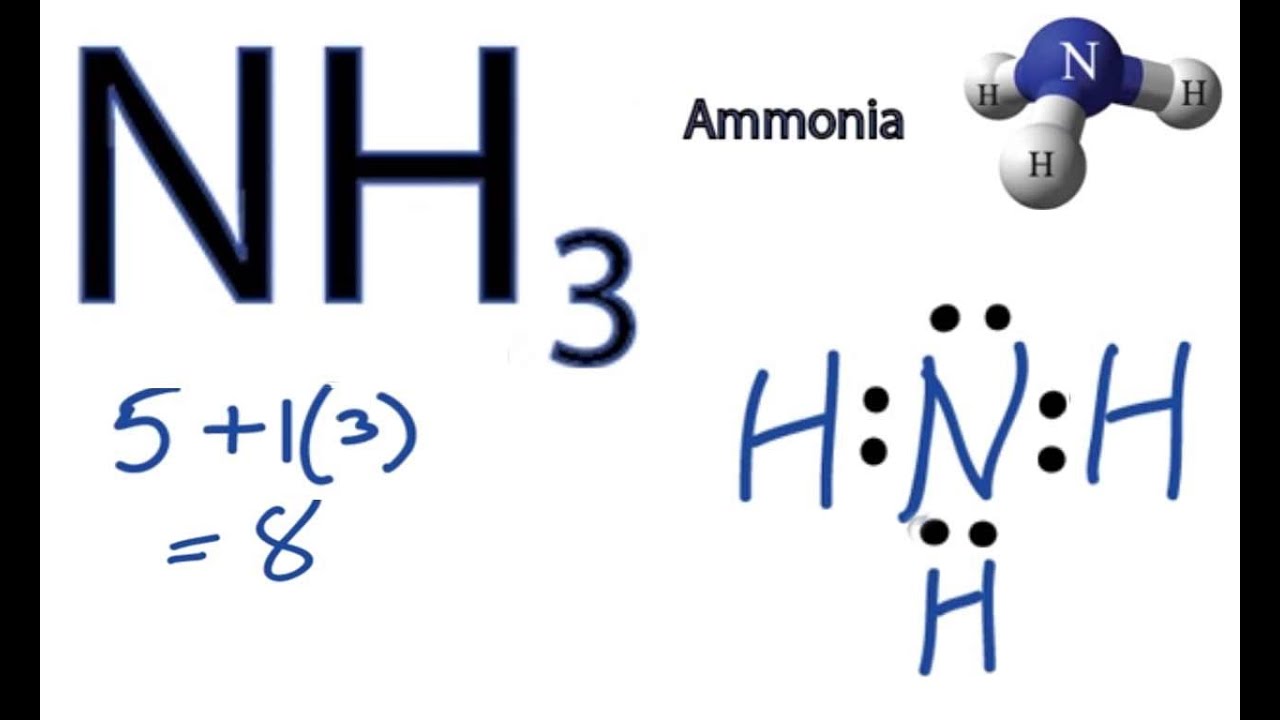








0 Response to "40 lewis dot diagram of ammonia"
Post a Comment